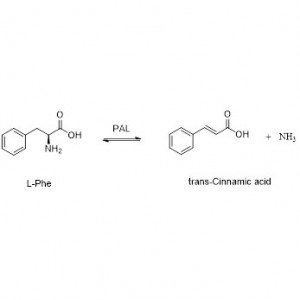
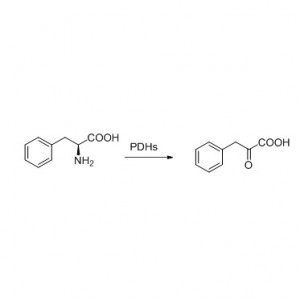
Amidasum (AMD)
Enzyma:Sunt catalysatores biologici macromoleculares, pleraque enzyma sunt proteina
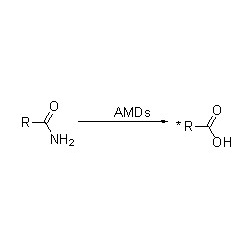
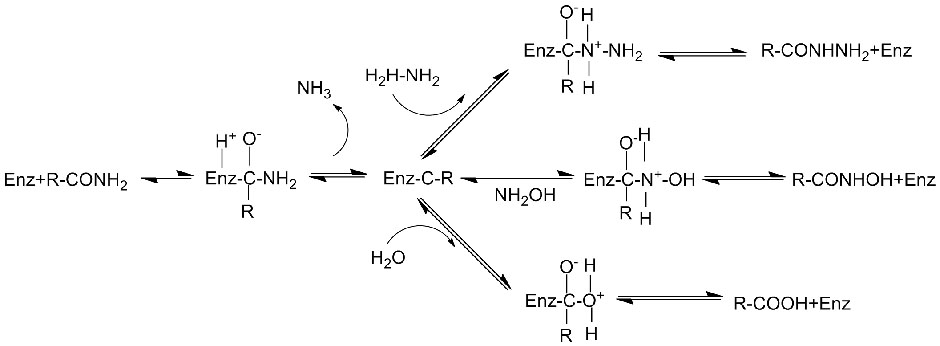
Amidasum:Hydrolysim variarum amidum aliphaticarum et aromaticarum, endogenarum et externarum, catalysare per translationem gregis acylici in aquam cum productione acidorum liberorum et ammoniae. Acida hydroxamica et alia acida organica late ut medicamenta adhibentur, quia constituentes factorum crescentium, antibioticorum et inhibitorum tumorum sunt. Amidases in acylases typi R et typi S dividi possunt secundum stereoselectivitatem catalysatoris.
Praeter hydrolysim amidum catalysandam, amidase etiam reactiones translationis acylicae in praesentia co-substratorum sicut hydroxylaminae catalysare potest.
Amidases cum diversis fontibus diversam substrati specificitatem habent, quarum quaedam tantum amida aromatica hydrolyzare possunt, quaedam tantum amida aliphatica hydrolyzare, et quaedam α-vel ω-aminoamida hydrolyzant. Pleraeque aminae bonam activitatem catalyticam tantum pro amidis aromaticis acyclicis vel simplicibus habent, sed pro aromaticis complexis, amidae heterocyclicae, praesertim amidae cum substituentibus ortho, plerumque activitate humili sunt (pauca tantum enzyma meliora effectus catalyticos exhibent).
Mechanismus catalyticus:
| Enzyma | Codex Producti | Codex Producti |
| Pulvis Enzymaticus | ES-AMD-101~ ES-AMD-119 | series 19 amidasarum, 50 mg singulae 19 res * 50mg / res, vel alia quantitas |
| Instrumentum Examinationis (SynKit) | ES-AMD-1900 | series 19 amidasarum, 1 mg singulae 19 res * 1 mg / res |
★ Alta substrati specificitas.
★ Selectivitas chiralis fortis.
★ Alta efficientia conversionis.
★ Pauciora producta secundaria.
★ Conditiones reactionis lenes.
★ Amicabilis ambienti.
➢ Examen enzymaticum pro substratis specificis propter specificitatem substrati peragendum est, et enzymum inveniendum est quod substratum destinatum cum effectu catalytico optimo catalysat.
➢ Numquam cum condicionibus extremis, ut puta: temperatura alta, pH altum/humile, et solvente organico altae concentrationis, contactum habe.
➢ Normaliter, systema reactionis substratum, solutionem tamponem (pH reactionis optimum enzymi) includere debet. Cosubstrata, ut hydroxylamina, in systemate reactionis translationis acylicae adesse debent.
➢ AMD ultimum in systema reactionis cum pH et temperatura reactionis optima addendum est.
➢ Omnia genera AMD varias condiciones optimas reactionis habent, itaque singula singillatim ulterius investiganda sunt.
Exemplum 1(1):
Actio hydrolytica ad varia substrata amidica
| Substratum | Actio specifica μmol/min-1mg-1 | Substratum | Actio specifica μmol/min-1mg-1 |
| Acetamidum | 3.8 | ο-OH benzamidum | 1.4 |
| Propionamidum | 3.9 | p-OH benzamidum | 1.2 |
| Lactamidum | 12.8 | ο-NH2benzamidum | 1.0 |
| Butyramidum | 11.9 | p-NH2benzamidum | 0.8 |
| Isobutyramidum | 26.2 | ο-Toluamidum | 0.3 |
| Pentanamidum | 22.0 | p-Toluamidum | 8.1 |
| Hexanamidum | 6.4 | Nicotinamidum | 1.7 |
| Cyclohexanamidum | 19.5 | Isonicotinamidum | 1.8 |
| Acrylamidum | 10.2 | Picolinamidum | 2.1 |
| Metacrylamidum | 3.5 | 3-Phenylpropionamidum | 7.6 |
| Prolinamidum | 3.4 | Indol-3-acetamidum | 1.9 |
| Benzamidum | 6.8 |
Reactio in solutione tamponata natrii phosphatis 50mM, pH 7.5, ad 70°C peracta est.
| Amidae | Hydroxylamina | Hydrazinum |
| Acetamidum | 8.4 | 1.4 |
| Propionamidum | 18.4 | 3.0 |
| Isobutyramidum | 25.0 | 22.7 |
| Benzamidum | 9.2 | 6.1 |
Reactio in solutione tamponata natrii phosphatis 50mM, pH 7.5, ad 70°C peracta est.
Concentrationes reagentium conexae: amida, 100 mM (benzamidum, 10 mM); hydroxylamina et hydrazinum, 400 mM; enzymum 0.9 μM.
Exemplum II(2):
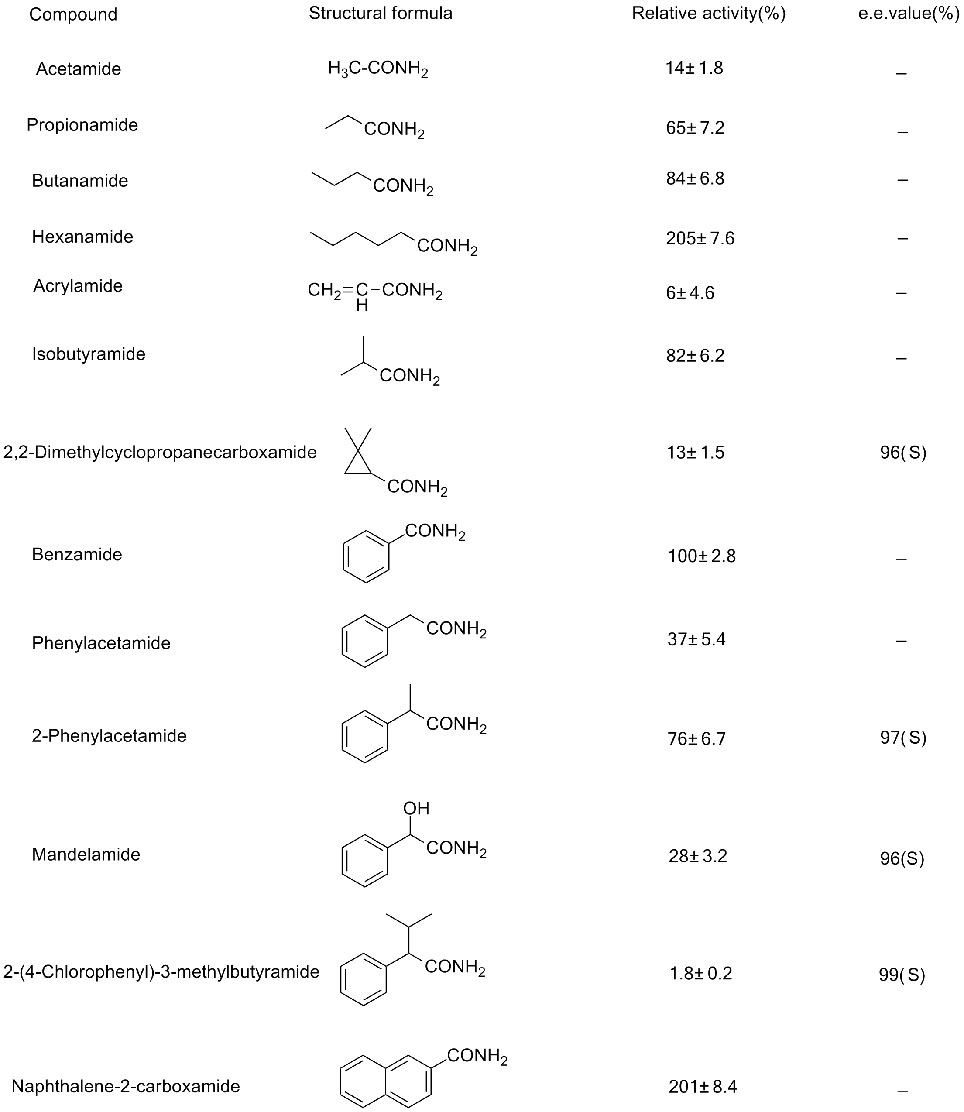
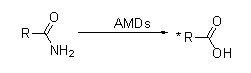
Exemplum III(3):
1. D'Abusco AS, Ammendola S., et al. Extremophiles, 2001, 5:183-192.
2. Guo FM, Wu JP, Yang LR, et al. *Processus Biochemistry*, 2015, 50(8): 1400-1404.
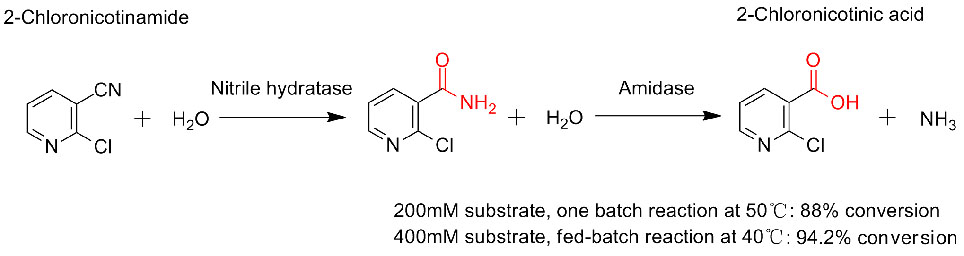
3. Zheng RC, Jin JQ, Wu ZM, et al. Chemia Bioorganica, 2017, In interreti praesto 7.